Abstract
Treatments for B-cell malignancies have improved over the past several decades with clinical application of the CD20-specific antibody rituximab and chimeric antigen receptor (CAR) T cells targeting CD19. Despite the success of these therapies, loss of CD20 after rituximab treatment has been reported in leukemia and lymphoma patients. Additionally, up to 50% of all patients receiving anti-CD19 CAR T-cell therapy relapse within the first year with many of those patients exhibiting CD19 loss. Thus, new therapeutic approaches are needed to address tumor antigen escape.
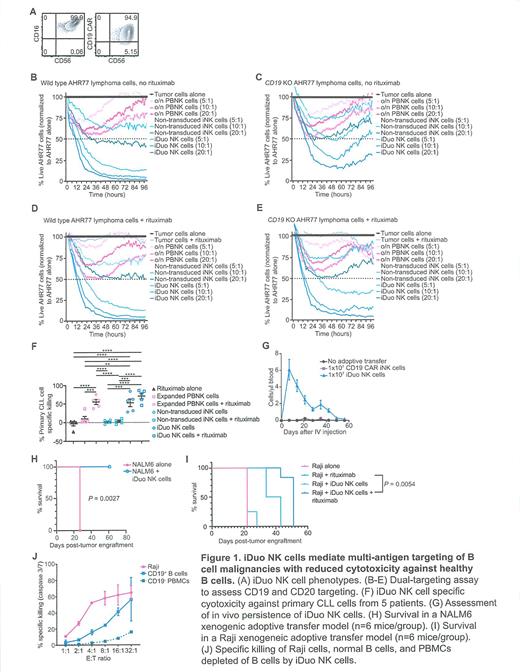
Accordingly, we generated triple gene-modified iPSC-derived NK (iNK) cells, termed "iDuo" NK cells, tailored to facilitate multi-antigen targeting. The iPSC line was clonally engineered to express high-affinity, non-cleavable CD16a (hnCD16), an anti-CD19 CAR optimized for NK cell signaling, and a membrane-bound IL-15/IL-15R fusion (IL-15RF) molecule to enhance NK cell persistence (Fig. 1A). To model antigen escape, we generated CD19 knockout AHR77 lymphoma cells alongside wild type AHR77 cells (both CD20 +) as targets in cytotoxicity assays. Activated peripheral blood NK (PBNK) cells, non-transduced iNK cells, and iDuo NK cells were tested as effectors. Unlike PBNK cells or non-transduced iNK cells, iDuo NK cells efficiently eliminated wild type AHR77 cells with or without the addition of rituximab at all tested E:T ratios. Similarly, iDuo NK cells in combination with rituximab were uniquely able to efficiently eliminate CD19 KO AHR77 cells due to enhanced antibody-dependent cellular cytotoxicity (ADCC) driven by hnCD16 (Fig. 1B-E). Cytotoxicity mediated by iDuo NK cells was also evaluated using primary chronic lymphocytic leukemia (CLL) cells. Compared to expanded PBNK cells and non-transduced iNK cells, only iDuo NK cells (in the absence of rituximab) were able to kill primary CLL cells (Fig. 1F).
Expression of IL-15RF by iDuo NK cells uniquely supports in vitro expansion without the need for cytokine supplementation. To determine whether IL-15RF supports in vivo persistence of iDuo NK cells, CD19 CAR iNK cells (lacking IL-15RF) and iDuo NK cells were injected into NSG mice without the addition of cytokines or CD19 antigen availability. iDuo NK cell numbers peaked within a week after injection and persisted at measurable levels for ~5 weeks, in marked contrast to CD19 CAR iNK cell numbers that were undetectable throughout (Fig. 1G).
To evaluate the in vivo function of iDuo NK cells, NALM6 leukemia cells were engrafted into NSG mice. Groups of mice received tumor alone or were treated with 3 doses of thawed iDuo NK cells. iDuo NK cells alone were highly effective in this model as evidenced by complete survival of mice in the treatment group (Fig. 1H). To assess iDuo NK cells in a more aggressive model, Raji lymphoma cells were engrafted, and groups of mice received rituximab alone, iDuo NK cells alone, or iDuo NK cells plus rituximab. Mice given the combination of iDuo NK cells and rituximab provided extended survival compared to all other arms in the aggressive disseminated Raji lymphoma xenograft model (Fig. 1I).
One disadvantage of anti-CD19 CAR T cells is their inability to discriminate between healthy and malignant B cells. Because NK cells express inhibitory receptors that enable "self" versus "non-self" discrimination, we reasoned that iDuo NK cells could have higher cytotoxicity against tumor cells relative to healthy B cells. To address this, we labeled Raji cells, CD19 + B cells from healthy donor peripheral blood mononuclear cells (PBMCs) and CD19 - PBMCs. Labeled populations of cells were co-cultured with iDuo NK cells, and specific killing was analyzed. As expected, iDuo NK cells did not target CD19 - PBMCs. Intriguingly, iDuo NK cells had much higher cytotoxic activity against Raji cells compared to primary CD19 + B cells, suggesting a preferential targeting of malignant B cells compared to healthy B cells.
Together, these results demonstrate the potent multi-antigen targeting capability and in vivo antitumor function of iDuo NK cells. Further, these data suggest that iDuo NK cells may have an additional advantage over anti-CD19 CAR T cells by discriminating between healthy and malignant B cells. The first iDuo NK cell, FT596, is currently being tested in a Phase I clinical trial (NCT04245722) for the treatment of B-cell lymphoma.
Cichocki: Gamida Cell: Research Funding; Fate Therapeutics, Inc: Patents & Royalties, Research Funding. Bjordahl: Fate Therapeutics: Current Employment. Gaidarova: Fate Therapeutics, Inc: Current Employment. Abujarour: Fate Therapeutics, Inc.: Current Employment. Rogers: Fate Therapeutics, Inc: Current Employment. Huffman: Fate Therapeutics, Inc: Current Employment. Lee: Fate Therapeutics, Inc: Current Employment. Szabo: Fate Therapeutics, Inc: Current Employment. Wong: BMS: Current equity holder in publicly-traded company; Fate Therapeutics, Inc: Current Employment. Cooley: Fate Therapeutics, Inc: Current Employment. Valamehr: Fate Therapeutics, Inc.: Current Employment. Miller: Magenta: Membership on an entity's Board of Directors or advisory committees; ONK Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees; Vycellix: Consultancy; GT Biopharma: Consultancy, Patents & Royalties, Research Funding; Fate Therapeutics, Inc: Consultancy, Patents & Royalties, Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees; Wugen: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal